京ICP备13020181号-2
© 《China Plastics》
© 《China Plastics》

China Plastics ›› 2023, Vol. 37 ›› Issue (11): 62-73.DOI: 10.19491/j.issn.1001-9278.2023.11.007
• Processing and Application • Previous Articles Next Articles
ZHANG Chao1, XU Shuangping1( ), JIA Hongge1, ZHANG Mingyu1, QU Yanqing1, XU Jingyu2
), JIA Hongge1, ZHANG Mingyu1, QU Yanqing1, XU Jingyu2
Received:2023-05-26
Online:2023-11-26
Published:2023-11-22
CLC Number:
ZHANG Chao, XU Shuangping, JIA Hongge, ZHANG Mingyu, QU Yanqing, XU Jingyu. Progress in preparation of graphene⁃like carbon nitride and its application in gas separation[J]. China Plastics, 2023, 37(11): 62-73.
| 方法名称 | 反应特点 | 材料性能影响 | 优缺点 | 参考文献 |
|---|---|---|---|---|
| 热聚合法 | 通过加热将单体激发为单体自由基,从而进行自由基聚合 | g⁃C3N4可作为光催化剂,在可见光下表现出可回收的吸附和光催化活性 | 以富含氰胺结构的前驱体在半封闭体系中按恒定的升温速率即可得到目标产物,操作简单,原料来源广泛。缺点是大多数情况下需要添加辅助剂,不适合大规模生产 | [ |
| 溶剂热法 | 在密闭体系中以有机物作为溶剂,通过液⁃固相反应进行缩合 | 使产物结晶良好,可以获得不同尺寸的单晶,通过改变反应条件控制反应物分子自组装过程 | 有着操作简单易于控制、体系均匀性好、隔绝外界环境因素。缺点是需在高温高压的反应条件下进行且无法直接观察反应进程 | [ |
| 固相反应法 | 2种或2种以上固体表面发生化学反应,生成新的固体产物 | 固相反应能控制反应物的结构,可以得到特殊形貌的 g⁃C3N4,例如得到纳米管、纳米线和假立方体等形貌的 g⁃C3N4 | 固相反应不需要溶剂,没有溶剂挥发和废液排放,降低了污染。缺点是反应物的颗粒尺寸和分散程度会影响反应的速率且反应时间较长,对反应条件的控制要求较高 | [ |
| 化学气相沉积法 | 气态的反应物或粉尘气流在一定温度和常压或真空条件下的密闭容器内发生反应,生成固体产物沉积在衬底上 | 通过控制反应时间控制气相沉积涂层的厚度,温度的改变也会影响反应物的形貌(颗粒状或薄膜等) | 反应物之间有着更大的接触面积,反应充分且速率快,可以获得平滑的沉积面。缺点是反应温度过高,导致基体材料的选择受到限制 | [ |
| 方法名称 | 反应特点 | 材料性能影响 | 优缺点 | 参考文献 |
|---|---|---|---|---|
| 热聚合法 | 通过加热将单体激发为单体自由基,从而进行自由基聚合 | g⁃C3N4可作为光催化剂,在可见光下表现出可回收的吸附和光催化活性 | 以富含氰胺结构的前驱体在半封闭体系中按恒定的升温速率即可得到目标产物,操作简单,原料来源广泛。缺点是大多数情况下需要添加辅助剂,不适合大规模生产 | [ |
| 溶剂热法 | 在密闭体系中以有机物作为溶剂,通过液⁃固相反应进行缩合 | 使产物结晶良好,可以获得不同尺寸的单晶,通过改变反应条件控制反应物分子自组装过程 | 有着操作简单易于控制、体系均匀性好、隔绝外界环境因素。缺点是需在高温高压的反应条件下进行且无法直接观察反应进程 | [ |
| 固相反应法 | 2种或2种以上固体表面发生化学反应,生成新的固体产物 | 固相反应能控制反应物的结构,可以得到特殊形貌的 g⁃C3N4,例如得到纳米管、纳米线和假立方体等形貌的 g⁃C3N4 | 固相反应不需要溶剂,没有溶剂挥发和废液排放,降低了污染。缺点是反应物的颗粒尺寸和分散程度会影响反应的速率且反应时间较长,对反应条件的控制要求较高 | [ |
| 化学气相沉积法 | 气态的反应物或粉尘气流在一定温度和常压或真空条件下的密闭容器内发生反应,生成固体产物沉积在衬底上 | 通过控制反应时间控制气相沉积涂层的厚度,温度的改变也会影响反应物的形貌(颗粒状或薄膜等) | 反应物之间有着更大的接触面积,反应充分且速率快,可以获得平滑的沉积面。缺点是反应温度过高,导致基体材料的选择受到限制 | [ |
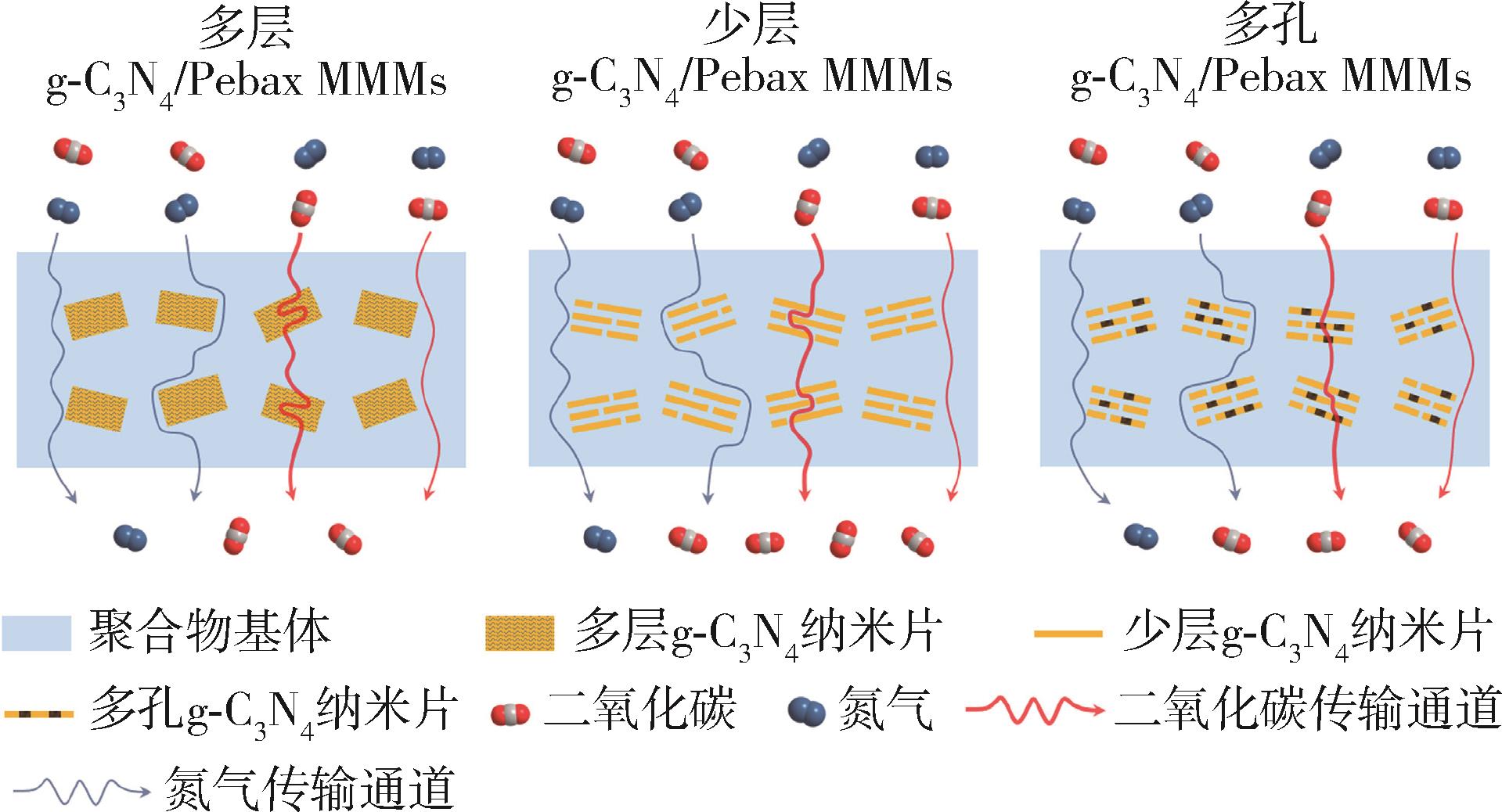
| 名称 | 应用特点 | 参考文献 |
|---|---|---|
| PIM⁃1/g⁃C3N4 | PIM (例如PIM⁃1等)具有较高的自由体积和优异的气体渗透性,热稳定性好结构简单;g⁃C3N4的加入能够合理调节混合基质膜的渗透性能,改善“trade⁃off”效应(渗透率提升导致选择性下降),可以有效影响PIM⁃1聚合物链的堆积,使得g⁃C3N4与PIM⁃1基体之间的界面产生额外的气体传输路径,从而提高渗透系数;g⁃C3N4具有筛分效应,周期性超微孔会促进H2的传输,提高了H2/CH4和H2/N2的选择性,并且使PIM⁃1/g⁃C3N4 MMMs表现出良好的长期性能;为在实际的H2净化、空气分离和CO2捕获中使用所开发的膜铺平了道路 | [ |
| Pebax/g⁃C3N4 | Pebax(例如Pebax⁃1657等)是一种价格低廉、易于获取的膜材料,被广泛用于气体分离膜领域;Pebax⁃1657具有很好的成膜性,能够耐受酸、碱和部分有机溶剂,有很强的实用性;g⁃C3N4显示出了CO2吸附和筛分性能,与纯Pebax膜相比,具有Pebax/g⁃C3N4 MMMs 在CO2渗透率和CO2/N2选择性方面同时得到了提高。此外,该类膜在长期运行测试中保持了其分离性能,显示了CO2捕获的巨大潜力,验证了其在气体净化工业中的应用潜力 | [ |
| g⁃C3N4 SILM | IL (例如1⁃乙基-3⁃甲基咪唑乙酸酯等)对CO2有着良好的选择吸收性能,离子液体支撑液膜是将多孔支撑材料浸在离子液体中,在表面张力作用下,离子液体充满微孔后形成,是一种新型的气体分离膜技术;g⁃C3N4 SILM能够高效分离CO2/N2和CO2/CH4,并且具有出色的稳定性和耐用性,为制备具有高气体分离性能的CO2分离膜开辟了一条简单的途径 | [ |
| PI/g⁃C3N4 | PI膜的力学性能好、热稳定性高,分离性尤为突出,被广泛用于气体分离膜领域;g⁃C3N4质子化改性后与PI之间存在较强的界面相互作用,使得杂化纳米复合膜具有较高的抗膨胀性能和力学稳定性,与纯PI膜相比,PI/g⁃C3N4 MMMs的CO2/CH4选择性增强 | [ |
| PES/g⁃C3N4 | PES 膜有稳定性强,耐酸碱程度高,耐热性非常好;CS改性的g⁃C3N4结合的ZIF⁃8 PES膜具有优异的CO2/CH4理想选择性(24.2),并且使膜的柔韧性提高 | [ |
| 名称 | 应用特点 | 参考文献 |
|---|---|---|
| PIM⁃1/g⁃C3N4 | PIM (例如PIM⁃1等)具有较高的自由体积和优异的气体渗透性,热稳定性好结构简单;g⁃C3N4的加入能够合理调节混合基质膜的渗透性能,改善“trade⁃off”效应(渗透率提升导致选择性下降),可以有效影响PIM⁃1聚合物链的堆积,使得g⁃C3N4与PIM⁃1基体之间的界面产生额外的气体传输路径,从而提高渗透系数;g⁃C3N4具有筛分效应,周期性超微孔会促进H2的传输,提高了H2/CH4和H2/N2的选择性,并且使PIM⁃1/g⁃C3N4 MMMs表现出良好的长期性能;为在实际的H2净化、空气分离和CO2捕获中使用所开发的膜铺平了道路 | [ |
| Pebax/g⁃C3N4 | Pebax(例如Pebax⁃1657等)是一种价格低廉、易于获取的膜材料,被广泛用于气体分离膜领域;Pebax⁃1657具有很好的成膜性,能够耐受酸、碱和部分有机溶剂,有很强的实用性;g⁃C3N4显示出了CO2吸附和筛分性能,与纯Pebax膜相比,具有Pebax/g⁃C3N4 MMMs 在CO2渗透率和CO2/N2选择性方面同时得到了提高。此外,该类膜在长期运行测试中保持了其分离性能,显示了CO2捕获的巨大潜力,验证了其在气体净化工业中的应用潜力 | [ |
| g⁃C3N4 SILM | IL (例如1⁃乙基-3⁃甲基咪唑乙酸酯等)对CO2有着良好的选择吸收性能,离子液体支撑液膜是将多孔支撑材料浸在离子液体中,在表面张力作用下,离子液体充满微孔后形成,是一种新型的气体分离膜技术;g⁃C3N4 SILM能够高效分离CO2/N2和CO2/CH4,并且具有出色的稳定性和耐用性,为制备具有高气体分离性能的CO2分离膜开辟了一条简单的途径 | [ |
| PI/g⁃C3N4 | PI膜的力学性能好、热稳定性高,分离性尤为突出,被广泛用于气体分离膜领域;g⁃C3N4质子化改性后与PI之间存在较强的界面相互作用,使得杂化纳米复合膜具有较高的抗膨胀性能和力学稳定性,与纯PI膜相比,PI/g⁃C3N4 MMMs的CO2/CH4选择性增强 | [ |
| PES/g⁃C3N4 | PES 膜有稳定性强,耐酸碱程度高,耐热性非常好;CS改性的g⁃C3N4结合的ZIF⁃8 PES膜具有优异的CO2/CH4理想选择性(24.2),并且使膜的柔韧性提高 | [ |
| 1 | 张娅, 王锐, 文思斯, 等. 石墨相氮化碳纳米片膜研究进展[J]. 化工学报, 2021, 72(12): 6 188⁃6 202. |
| ZHANG Y, WANG R, WEN S S,et al.Research progress of graphitic carbon nitride nanosheets membrane[J]. CIESC Journal, 2021, 72(12): 6 188⁃6 202. | |
| 2 | Liebig J. Uber einige Stickstoff‐Verbindungen[J]. Annalen der Pharmacie, 1834, 10(1): 1⁃47. |
| 3 | Franklin E C. The ammono carbonic acids[J]. Journal of the American Chemical Society, 1922, 44(3): 486⁃509. |
| 4 | Pauling L, Sturdivant J H. The structure of cyameluric acid, hydromelonic acid and related substances[J]. Proceedings of the National Academy of Sciences, 1937, 23(12): 615⁃620. |
| 5 | Liu A Y, Cohen M L. Prediction of new low compressibility solids[J]. Science, 1989, 245(4 920): 841⁃842. |
| 6 | Teter D M, Hemley R J. Low⁃compressibility carbon nitrides[J]. Science, 1996, 271(5 245): 53⁃55. |
| 7 | Chen Z, Lan Y H, Hong Y B, et al. Review of 2D graphitic carbon nitride⁃based membranes: principles, syntheses, and applications[J]. Applied Nano Materials, 2022, 5(9):12 343⁃12 365. |
| 8 | Goh S H, Lau H S, Yong W F. Metal⁃organic frameworks (MOFs)⁃based mixed matrix membranes (MMMs) for gas separation: a review on advanced materials in harsh environmental applications[J]. Small, 2022, 18(20):2107536. |
| 9 | 周进, 丁玲, 张婷, 等. g⁃C3N4 /CQDs光催化材料的制备及性能[J]. 精细化工, 2020, 4: 702⁃709. |
| ZHOU J, DING L, ZHANG T,et al.Preparation and properties of g⁃C3N4/CQDs photocatalyst materials[J]. Fine Chemicals, 2020,4: 702⁃709. | |
| 10 | 孙少峰, 涂琴, 张丽.CeO2/g⁃C3N4复合光催化剂的制备及其性能研究[J]. 水处理技术, 2021, 4: 52⁃55. |
| SUN S F, TU Q, ZHANG L.Study on the preparation and performance of CeO2/g⁃C3N4 composite photocatalyst[J]. Technology of Water Treatment, 2021, 4: 52⁃55. | |
| 11 | Kroke E, Schwarz M, Horath⁃Bordon E, et al. Tri⁃s⁃triazine derivatives. Part I. From trichloro⁃tri⁃s⁃triazine to graphitic C3N4 structures[J]. New Journal of Chemistry, 2002, 26: 508⁃512. |
| 12 | Ma X G, Lv Y H, Xu J, et al. A strategy of enhancing the photoactivity of g⁃C3N4 via doping of nonmetal elements: a first⁃principles study[J]. The Journal of Physical Chemistry C, 2012, 116(44): 23 485⁃23 493. |
| 13 | Wang Y, Wang X C, Antonietti M. Polymeric graphitic carbon nitride as a heterogeneous organocatalyst: from photochemistry to multipurpose catalysis to sustainable chemistry[J]. Angewandte Chemie International Edition, 2012, 51(1): 68⁃89. |
| 14 | Cao S, Chen H, Jiang F, et al. Nitrogen photofixation by ultrathin amine⁃functionalized graphitic carbon nitride nanosheets as a gaseous product from thermal polymerization of urea[J]. Applied Catalysis B: Environmental, 2018, 224: 222⁃229. |
| 15 | Liu J, Zhang T, Wang Z, et al. Simple pyrolysis of urea into graphitic carbon nitride with recyclable adsorption and photocatalytic activity[J]. Journal of Materials Chemistry, 2011, 21(38): 14 398⁃14 401. |
| 16 | Sun C, Dai J, Zhang H, et al. A facile method for preparing porous g⁃C3N4 nanosheets with efficient photocatalytic activity under visible light[J]. Journal of Materials Science, 2021, 56(12): 7 557⁃7 572. |
| 17 | Yan S C, Li Z S, Zou Z G. Photodegradation performance of g⁃C3N4 fabricated by directly heating melamine[J]. Langmuir, 2009, 25(17): 10 397⁃10 401. |
| 18 | Zhai H S, Cao L, Xia X H. Synthesis of graphitic carbon nitride through pyrolysis of melamine and its electrocatalysis for oxygen reduction reaction[J]. Chinese Chemical Letters, 2013, 24(2): 103⁃106. |
| 19 | Zhou C, Shi R, Shang L, et al. Template⁃free large⁃scale synthesis of g⁃C3N4 microtubes for enhanced visible light⁃driven photocatalytic H2 production[J]. Nano Research, 2018, 11(6): 3 462⁃3 468. |
| 20 | Zhang Y, Gong H, Li G, et al. Synthesis of graphitic carbon nitride by heating mixture of urea and thiourea for enhanced photocatalytic H2 production from water under visible light[J]. International Journal of Hydrogen Energy, 2017, 42(1): 143⁃151. |
| 21 | Qin Y, Ding Y, Tang H. Highly efficient visible⁃light photocatalytic activity of graphitic carbon nitride prepared from melamine⁃thiourea molecular composite[J]. Journal of Environmental Chemical Engineering, 2016, 4(4): 4 374⁃4 384. |
| 22 | ZHANG J S, WANG B, WANG X C. Chemical synthesis and applications of graphitic carbon nitride[J]. Acta Physico⁃Chimica Sinica, 2013, 29(9): 1 865⁃1 876. |
| 23 | Montigaud H, Tanguy B, Demazeau G, et al. Solvothermal synthesis of the graphitic form of C3N4 as macroscopic sample[J]. Diamond and related Materials, 1999, 8(8/9): 1 707⁃1 710. |
| 24 | Montigaud H, Tanguy B, Demazeau G, et al. C3N4: Dream or reality? Solvothermal synthesis as macroscopic samples of the C3N4 graphitic form[J]. Journal of materials science, 2000, 35(10): 2 547⁃2 552. |
| 25 | Bai Y J, Lü B, Liu Z G, et al. Solvothermal preparation of graphite⁃like C3N4 nanocrystals[J]. Journal of crystal growth, 2003, 247(3/4): 505⁃508. |
| 26 | Luv Q, Cao C B, Zhang J T, et al. Benzene thermal synthesis and characterization of crystalline carbon nitride[J]. Applied Physics A, 2004, 79(3): 633⁃636. |
| 27 | Andreyev A, Akaishi M, Golberg D. Sodium flux⁃assisted low⁃temperature high⁃pressure synthesis of carbon nitride with high nitrogen content[J]. Chemical physics letters, 2003, 372(5/6): 635⁃639. |
| 28 | Li J, Cao C, Hao J, et al. Self⁃assembled one⁃dimensional carbon nitride architectures[J]. Diamond & Related Materials, 2006, 15(10): 1 593⁃1 600. |
| 29 | Bai X, Li J, Cao C, et al. Solvothermal synthesis of the special shape (deformable) hollow g⁃C3N4 nanospheres[J]. Materials Letters, 2011, 65(7): 1 101⁃1 104. |
| 30 | Guo Q, Yang Q, Yi C, et al.Synthesis of carbon nitrides with graphite⁃like or onion⁃like lamellar structures via a solvent⁃free route at low temperatures[J]. Carbon,2005,43(7): 1 386⁃1 391. |
| 31 | Gu Y, Chen L, Shi L, et al. Synthesis of C3N4 and graphite by reacting cyanuric chloride with calcium cyanamide[J]. Carbon, 2003, 41(13): 2 674⁃2 676. |
| 32 | Khabashesku V N, Zimmerman J L, Margrave J L. Powder synthesis and characterization of amorphous carbon nitride[J]. Chemistry of Materials, 2000, 12(11): 3 264⁃3 270. |
| 33 | Zhang Z, Leinenweber K, Bauer M, et al. High pressure bulk synthesis of crystalline C6N9H3·HCl a new compound containing a layered C⁃N network[J]. Journal of the American Chemical Society, 2001, 123(32): 7 788⁃7 796. |
| 34 | Li Y, Zhang J, Wang Q, et al. Nitrogen⁃rich carbon nitride hollow vessels: synthesis, characterization, and their properties[J]. The Journal of Physical Chemistry B, 2010, 114(29): 9 429⁃9 434. |
| 35 | Tragl S, Gibson K, Glaser J, et al. Template assisted formation of micro⁃and nanotubular carbon nitride materials[J]. Solid State Communications, 2007, 141(9): 529⁃534. |
| 36 | Zimmerman J L, Williams R, Khabashesku V N, et al. Synthesis of spherical carbon nitride nanostructures[J]. Nano Letters, 2001, 1(12): 731⁃734. |
| 37 | Lu X, Gai L, Cui D, et al. Synthesis and characterization of C3N4 nanowires and pseudocubic C3N4 polycrystalline nanoparticles[J]. Materials Letters, 2007, 61(21): 4 255⁃4 258. |
| 38 | Tanaka I, Sakamoto Y. Low⁃temperature synthesis of carbon nitride by microwave plasma CVD[J]. Japanese Journal of Applied Physics, 2015, 55(1): 01AA15. |
| 39 | Kosaka M, Urakami N, Hashimoto Y. Formation of graphitic carbon nitride and boron carbon nitride film on sapphire substrate[J]. Japanese Journal of Applied Physics, 2018, 57(2S2): 02CB09. |
| 40 | Yadav R M, Kumar R, Aliyan A, et al. Facile synthesis of highly fluorescent free⁃standing films comprising graphitic carbon nitride (g⁃C3N4) nanolayers[J]. New Journal of Chemistry, 2020, 44(6): 2 644⁃2 651. |
| 41 | Chamorro⁃Posada P, Dante R C, Cabo J V, et al. Experimental and theoretical investigations on a CVD grown thin film of polymeric carbon nitride and its structure[J]. Diamond and Related Materials, 2020,111: 108169. |
| 42 | Chubenko E B, Kovalchuk N G, Komissarov I V, et al. Chemical vapor deposition of 2D crystallized g⁃C3N4 layered films[J]. The Journal of Physical Chemistry C, 2022, 126(9): 4 710⁃4 714. |
| 43 | Tian Z, Wang S, Wang Y, et al. Enhanced gas separation performance of mixed matrix membranes from graphitic carbon nitride nanosheets and polymers of intrinsic microporosity[J]. Journal of Membrane Science, 2016, 514: 15⁃24. |
| 44 | Voon B K, Lau H S, Liang C Z, et al. Functionalized two⁃dimensional g⁃C3N4 nanosheets in PIM-1 mixed matrix membranes for gas separation[J]. Separation and Purification Technology, 2022: 121354. |
| 45 | Cheng L, Song Y, Chen H, et al. g⁃C3N4 nanosheets with tunable affinity and sieving effect endowing polymeric membranes with enhanced CO2 capture property[J]. Separation and Purification Technology, 2020, 250: 117200. |
| 46 | Guo F, Li D, Ding R, et al. Constructing MOF⁃doped two⁃dimensional composite material ZIF-90@C3N4 mixed matrix membranes for CO2/N2 separation[J]. Separation and Purification Technology, 2022, 280: 119803. |
| 47 | Chen D, Ying W, Guo Y, et al. Enhanced gas separation through nanoconfined ionic liquid in laminated MoS2 membrane[J]. ACS applied materials & interfaces, 2017, 9(50): 44 251⁃44 257. |
| 48 | Ying W, Hou Q, Chen D, et al. Electrical field facilitates selective transport of CO2 through a laminated MoS2 supported ionic liquid membrane[J]. Journal of Materials Chemistry A, 2019, 7(16): 10 041⁃10 046. |
| 49 | Niu Z, Luo W, Mu P. Nanoconfined CO2⁃philic ionic liquid in laminated g⁃C3N4 membrane for the highly efficient separation of CO2 [J]. Separation and Purification Technology, 2022, 297: 121593. |
| 50 | Soto⁃Herranz M, Sánchez⁃Báscones M, Hérnandez⁃Giménez A, et al. Effects of protonation, hydroxylamination, and hydrazination of g⁃C3N4 on the performance of matrimid®/g⁃C3N4 membranes[J]. Nanomaterials, 2018, 8(12): 1010. |
| 51 | Jomekian A, Bazooyar B, Esmaeilzadeh J, et al. Highly CO2 selective chitosan/g⁃C3N4/ZIF-8 membrane on polyethersulfone microporous substrate[J]. Separation and Purification Technology, 2020, 236: 116307. |
| 52 | 郭海燕, 彭东来, 冯孝权, 等. 自具微孔聚合物PIM⁃1膜在气体分离领域的研究进展[J]. 化工进展, 2021, 40(10): 5 577⁃5 589. |
| GUO H Y, PENG D L, FENG X Q,et al.Progress in the membranes of polymers of intrinsic micro⁃porosity PIM-1 for gas separation[J]. Chemical Industry and Engineering Progress, 2021, 40(10): 5 577⁃5 589. | |
| 53 | 沈江南, 阮慧敏, 吴东柱, 等. 离子液体支撑液膜的研究及应用进展[J]. 化工进展, 2009,12: 2 092⁃2 098. |
| SHEN J N, RUAN H M, WU D Z,et al.Progress of supported liquid membrane with ionic liquids[J]. Chemical Industry and Engineering Progress, 2009,12: 2 092⁃2 098. |
| [1] | LI Dan, ZHAO Biao, CHEN Ke, WANG Fan, ZHANG Jingyu, ZHANG Fengbo, PAN Kai. Research progress in preparation and applications of hollow polypropylene fiber [J]. China Plastics, 2023, 37(9): 109-114. |
| [2] | YANG Xiaolong, CHEN Wenjing, LI Yongqing, YAN Xiaokun, WANG Xiulei, XIE Pengcheng, MA Xiuqing. Research progress in polymer/graphene conductive composites [J]. China Plastics, 2022, 36(6): 165-173. |
| [3] | WANG Rongchen, ZHANG Heng, SUN Huanwei, DUAN Shuxia, QIN Zixuan, LI Han, ZHU Feichao, ZHANG Yifeng. Research progress in preparation and hydrophilic modification of polylactic acid nonwovens for medical and health applications [J]. China Plastics, 2022, 36(5): 158-166. |
| [4] | LIU Wen, SHI Wenzhao, LIU Jinshu, LU Shaofeng, ZHOU Hongjuan. Research progress in electro⁃active shape memory composite materials [J]. China Plastics, 2022, 36(4): 175-189. |
| [5] | LI Yushan, LI Jie. Research progress in durable super⁃hydrophobic surface based on PDMS [J]. China Plastics, 2022, 36(3): 167-176. |
| [6] | LIU Yankuan, GU Zichen, WANG Zhiping. Preparation technology and development trend of continuous⁃fiber⁃reinforced thermoplastic prepregs [J]. China Plastics, 2022, 36(2): 172-181. |
| [7] | LI Ningli, WANG Meng, WANG Rui, ZHU Zhuangzhuang. Study on preparation technology of rubber⁃plastic alloy modified asphalt [J]. China Plastics, 2022, 36(12): 78-85. |
| [8] | QU Yanqing, ZHA Yuxin, JIA Hongge, XU Shuangping, ZHANG Mingyu. Research progress in preparation of separation membranes using interfacial polymerization technology [J]. China Plastics, 2022, 36(11): 150-157. |
| [9] | ZHANG Zhouya, BAI Shijian, ZHANG Yuxia, ZHOU Hongfu, GONG Fangfang, TANG Xueguli, WANG Bin. Research Progress in Factors Affecting Thermal Conductivity of Polymeric Materials [J]. China Plastics, 2021, 35(9): 156-165. |
| [10] | MA Chaoqun, SHI Wenzhao, CUI Shanshan, ZHANG Manyan, ZHOU Hongjuan. Research Progress in Polyvinyl Alcohol⁃based Porous Composites [J]. China Plastics, 2021, 35(4): 116-123. |
| [11] | YANG Xiaolong, LI Yongqing, YAN Xiaokun, CHEN Wenjing, MA Xiuqing. Research progress in gas barrier polymer/graphene composites [J]. China Plastics, 2021, 35(12): 145-153. |
| [12] | FANG Yunfeng, MA Biao, WANG Xiaoqing, KANG Xingxiang, TANG Yuting. Research progress in latent curing agent for one⁃component epoxy resin [J]. China Plastics, 2021, 35(12): 154-165. |
| [13] | Xiang YU, Huanhuan TAN, Zhuo WANG, Yukun ZHANG, Yanfei AN, Junjie ZHAO. Preparation and Performance Investigation of PA6/Aerogel Composite Powders with Phase Change Function by Solvent Precipitation Method [J]. China Plastics, 2020, 34(8): 71-77. |
| [14] | WEI Shiyi, TANG Yuntao, CHAI Chenze, ZHANG Yuxia, ZHOU Hongfu. Research Progress in Poly(lactic acid) Open⁃Cell Materials [J]. China Plastics, 2020, 34(10): 100-109. |
| [15] | . Research Progresses in Branched Polyamide 6 [J]. China Plastics, 2017, 31(08): 1-6 . |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||