 京公网安备11010802034965号
京ICP备13020181号-2
京公网安备11010802034965号
京ICP备13020181号-2
《中国塑料》编辑部 ©2008-2024 版权所有
地址:北京市海淀区阜成路11号 邮编:100048
编辑部:010-68985541 联系信箱:cp@plaschina.com.cn
广告部/发行部:010-68985253 本系统由北京玛格泰克科技发展有限公司设计开发

中国塑料 ›› 2024, Vol. 38 ›› Issue (1): 117-123.DOI: 10.19491/j.issn.1001-9278.2024.01.017
• 综述 • 上一篇
收稿日期:2023-08-17
出版日期:2024-01-26
发布日期:2024-01-22
通讯作者:
朱光明(1963-),男,教授,主要从事热、电、磁、光敏多相结构形状记忆聚合物的制备及性能研究,gmzhu@nwpu.edu.cn
GUO Xinyi, ZHU Guangming( ), ZHANG Xuebei, LI Ruichao
), ZHANG Xuebei, LI Ruichao
Received:2023-08-17
Online:2024-01-26
Published:2024-01-22
Contact:
ZHU Guangming
E-mail:gmzhu@nwpu.edu.cn
摘要:
归纳分析了三氟甲基类卡宾化合物、重氮酯类卡宾化合物、N⁃杂化卡宾化合物等稳定结构的卡宾前驱体的结构、性能特点,并重点介绍了其在聚合物改性领域的应用研究进展。
中图分类号:
郭欣怡, 朱光明, 张雪蓓, 李锐超. 卡宾在聚合物交联反应中的研究进展[J]. 中国塑料, 2024, 38(1): 117-123.
GUO Xinyi, ZHU Guangming, ZHANG Xuebei, LI Ruichao. Research progress in Carbene in crosslinking reactions of polymers[J]. China Plastics, 2024, 38(1): 117-123.
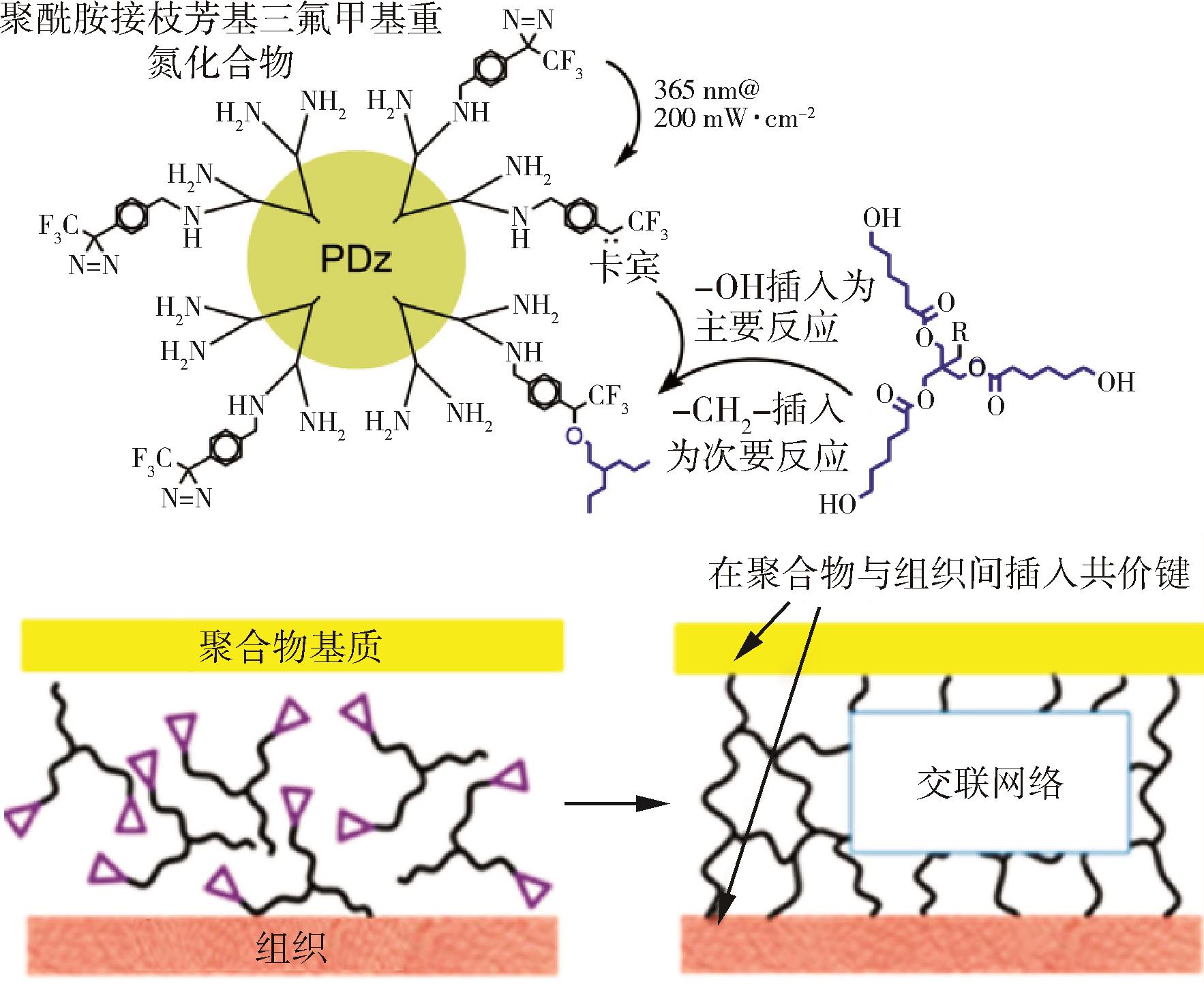
| 1 | Bourissou D, Guerret O, Francois P, et al. Stable carbenes [J] . Chemical Reviews, 2000(1):100. |
| 2 | Meerwein, Rathien H, Wemer H, Ber. Die methylierung von RH⁃verbindungen mittels diazomethas tnter mitirkung des lichtes[J]. Desch.Chem.Ges.1942,75(12):1 610⁃1 622. |
| 3 | Barton L, Nicholls D. Proceedings of the chemical society(London)[N].1964:443⁃444. |
| 4 | Liu Lijian, Ying Song, Hao Li. Carbene polymerization: characterization of poly(carballyloxycarbene)[J]. Polym Int,2002,51:1 047. |
| 5 | Noels A. Carbene chemistry: stereoregular polymers from diazo compounds[J]. Angewandte Chemie International Edition, 2007,46:1 208. |
| 6 | Jurberg Igor D, M L Davies Huw. Blue light⁃promoted photolysis of aryldiazoacetates[J]. Chem Sci,2018,9(22):5 112⁃5 118. |
| 7 | Bleiziffer Alexander, Kost Jonas, Rühe Jurgen. Thermal structuring of surface⁃attached polymer networks by C,H insertion reactions[J]. Macromol Mater Eng,2022,307:2 200. |
| 8 | Prucker O, Brandstetter T, Rühe, Jürgen. Surface⁃attached hydrogel coatings via C,H⁃insertion crosslinking for biomedical and bioanalytical applications (review) [J]. Biointerphases, 2018, 13(1):010801. |
| 9 | Prucker O, Brandstetter T, Rühe, Jürgen. Surface⁃attached hydrogel coatings via C,H⁃insertion crosslinking for biomedical and bioanalytical applications[J]. Biointerphases, 2018, 13(1):010801. |
| 10 | Aviv I, Gross Z. Iron(III) corroles and porphyrins as superior catalysts for the reactions of diazoacetates with nitrogen⁃ or sulfur⁃containing nucleophilic substrates: synthetic uses and mechanistic insights[J]. Chemistry⁃A European Journal, 2010, 14(13):3 815⁃4 123. |
| 11 | Brunner J, Senn H, Richards F M. 3⁃Trifluoromethyl⁃3⁃phenyldiazirine. a new carbene generating group for photolabeling reagents[J]. Journal of Biological Chemistry, 1980, 255(8):3 313⁃3 318. |
| 12 | Sato S, Yamada M, Wakahara T, et al. Photo⁃labeling of C 60 with 3⁃trifluoromethyl-3⁃phenyldiazirine[J]. Tetrahedron Letters, 2007, 48(36):6 290⁃6 293. |
| 13 | Yamada M, Harada K, Maeda Y, et al. A versatile approach to functionalisation of [60]fullerene using 3⁃trifluoromethyl⁃3⁃phenyldiazirine derivatives as photolabelling reagents[J]. New Journal of Chemistry, 2013, 37(11):3 762⁃3 769. |
| 14 | Hatanaka, Yasumaru, Sadakane, et al. Photoaffinity electrophoretic mobility shift assay using photoreactive DNA bearing 3⁃trifluoromethyl-3⁃phenyldiazirine in its phosphate backbone[J]. Analytical Biochemistry, 2016, 506:1⁃7. |
| 15 | Wicaksono, Djordjevic Gautama, Shah Ivan, al et, Photorheology of bioadhesive dendrimer polycaprolactone composites[J]. Polymer Testing, 2019, 80:106099. |
| 16 | Id A, Op A, Ahs A, et al. CaproGlu: multifunctional tissue adhesive platform[J]. Biomaterials, 2020, 260:120215. |
| 17 | Djordjevic Ivan, Wicaksono Gautama, Solic Ivan, et al. In vitro biocompatibility of diazirine⁃grafted biomaterials[J]. Macromol. Rapid Commun, 2020, 41:2000235 . |
| 18 | Djordjevic Ivan, Wicaksono Gautama, Singh Juhi, et al. Hybrid polymer networks of carbene and thiol ene[J].European Polymer Journal, 2022, 178:111502. |
| 19 | Lepage M L, Simhadri C, Liu C, et al. A broadly applicable cross⁃linker for aliphatic polymers containing C—H bonds[J]. Science, 2019, 366(6467):875⁃878. |
| 20 | Simhadri Chakravarthi, Bi Liting, Lepage Mathieu L., et l. Flexible polyfluorinated bis⁃diazirines as molecular adhesives[J]. Chem Sci, 2021,12:4 147. |
| 21 | Mathieu L, Lepage, Takaffoli Mahdi, Simhadri Chakravarthi, et al. Influence of topical cross⁃linking on mechanical and ballistic performance of a woven ultra⁃high⁃molecular⁃weight polyethylene fabric used in soft body armor[J]. Polym Mater, 2021, 3:6 008⁃6 018. |
| 22 | Musolino S F, Mahbod M, Nazir R, et al. Electronically optimized diazirine⁃based polymer crosslinkers[J].Polymer Chemistry, 2022, 13: 3 833⁃3 839. |
| 23 | Musolino S F, Pei Z, Bi L, et al. Structure⁃function relationships in aryl diazirines reveal optimal design features to maximize C—H insertion[J]. Chemical Science, 2021, 12:12138. |
| 24 | Zhao Xiaoxiao, Bi Liting, Khatir Behrooz, et al. Crosslinking inert liquidlike polydimethylsiloxane brushes using bis⁃diazirine chemical insertion for enhanced mechanical durability[J]. Chemical Engineering Journal, 2022, 442:136017. |
| 25 | Dey K, Chowdhury S R, Dykstra E, et al. Effect of bis⁃diazirine⁃mediated photo⁃crosslinking on polyvinylcarbazole and solution⁃processed polymer LEDs[J]. ACS Applied Electronic Materials, 2021, 3(8):3 365⁃3 371. |
| 26 | Ping Jianfeng, Feng Gao, Jian Lin Chen, et al. Adhesive curing through low⁃voltage activation[J]. Nature Communications, 2015, 6:8 050. |
| 27 | Pechmann H v.Berichte der deutschen chemischen Gesellschaft. ueber diazomethan und nitrosoacylamine[J].Chemistry Europe,1898,31(3):2 640⁃2 646. |
| 28 | Kotrade Philip F, Rghe Jgrgen. Malonic acid diazoesters for C⁃H insertion crosslinking (CHic) reactions: a versatile method for the generation of tailor⁃made surfaces[J]. Angew Chem Int Ed, 2017, 56:14 405⁃14 410. |
| 29 | Osterwinter G J, Navarro⁃Crespo R, Prucker O, et al. Surface‐attached polymer networks through carbene intermediates generated from α‐diazo esters[J]. Journal of Polymer Science Part A Polymer Chemistry, 2017:3 159⁃3 316. |
| 30 | Kost J, Bleiziffer A, Rusitov D, et al. Thermally induced cross⁃linking of polymers via C H insertion cross⁃linking (CHic) under mild conditions[J]. Journal of the American Chemical Society, 2021, 143(27):10 019⁃10 480. |
| 31 | Bleiziffer Alexander, Kost Jonas, Rühe Jurgen. Thermal structuring of surface⁃attached polymer networks by C H insertion reactions[J]. Macromol Mater Eng,2022,307:2 200. |
| 32 | Yang Shicheng, Yi Siyu, Yun Jie, et al. Carbene⁃mediated polymer cross⁃linking with diazo compounds by C—H activation and insertion[J]. Macromolecules, 2022, 55 (9):3 423⁃3 429. |
| 33 | Yi Yuan⁃Qiu⁃Qiang, Qi Dawei, Wei Honghui, et al. Molecular design of diazo compound for carbene⁃mediated cross⁃linking of hole⁃transport polymer in QLED with reduced energy barrier and improved charge balance[J]. ACS Appl Mater Interfaces, 2022, 14: 39 149⁃39 158. |
| 34 | Romain L, Anne⁃Laure W, Sofiem G, et al. Pd(II)–NHC coordination⁃driven formation of water⁃soluble catalytically active single chain nanoparticles[J]. Polymer Chemistry, 2018, 9(10):3 199⁃3 204. |
| 35 | Lambert R, Wirotius A L, Vignolle J, et al. C–C couplings in water by micellar catalysis at low loadings from a recyclable polymer⁃supported Pd(II)⁃NHC nanocatalyst[J]. Polymer Chemistry, 2018, 10(4):460⁃466. |
| 36 | Mehenni H, Pourcelle V, Gohy J F, et al. Synthesis and application of new photocrosslinkers for poly(ethylene glycol) [J]. Australian Journal of Chemistry, 2012, 65(2):193⁃201. |
| 37 | Jing L, Moloney M G, Xu H, et al. Carbene modification and reversible crosslinking of silver nanoparticles for controlled antibacterial activity[J]. Scientific Reports, 2020, 10(1):14 937. |
| [1] | 任国振, 王蒙蒙, 黄建建, 晋刚. 体积拉伸流场下PEEK/TLCP共混物的制备及性能研究[J]. 中国塑料, 2023, 37(8): 1-7. |
| [2] | 吕冲, 朱雯雯, 史正雪, 加英姿, 江学良, 游峰, 黄李纲, 姚楚, 刘仿军. 高降噪聚合物基复合材料的制备及声学性能研究进展[J]. 中国塑料, 2023, 37(11): 170-177. |
| [3] | 高永红, 陈凌峰, 金清平. 冻融环境下GFRP管混凝土柱轴压性能试验研究[J]. 中国塑料, 2023, 37(1): 74-81. |
| [4] | 高永红, 彭梦蜜, 金清平. 温度对玻璃纤维增强聚合物筋与混凝土黏结性能影响试验研究[J]. 中国塑料, 2022, 36(9): 16-23. |
| [5] | 杨超永, 郭金强, 王富玉, 张玉霞. 高性能塑料薄膜制备方法及改性研究进展[J]. 中国塑料, 2022, 36(9): 167-179. |
| [6] | 焦志伟, 王克琛, 张杨, 杨卫民. 基于碳纳米涂层沉积滑石粉与炭黑协同填充PVC/ABS复合材料的性能研究[J]. 中国塑料, 2022, 36(8): 10-15. |
| [7] | 杨岩, 王杰, 李宗育, 王懿明, 王运楠, 黎水娟, 雷良才, 李海英. 超支化离子液体聚合物合成方法综述[J]. 中国塑料, 2022, 36(8): 159-165. |
| [8] | 杨小龙, 陈文静, 李永青, 闫晓堃, 王修磊, 谢鹏程, 马秀清. 导电型聚合物/石墨烯复合材料的研究进展[J]. 中国塑料, 2022, 36(6): 165-173. |
| [9] | 孙旗, 高兴, 崔雪梅, 连慧琴, 崔秀国, 汪晓东. 黑磷烯纳米阻燃剂研究进展[J]. 中国塑料, 2022, 36(5): 133-139. |
| [10] | 田驰锋, 张洪申. 基于翅片式摩擦桶的车用聚合物粒子荷电及静电分离探索[J]. 中国塑料, 2022, 36(5): 75-80. |
| [11] | 宋立健, 张有忱, 左夏华, 张政和, 安瑛, 杨卫民, 谭晶, 程礼盛. 自组装单分子层调控界面热输运的研究进展[J]. 中国塑料, 2022, 36(4): 60-69. |
| [12] | 程曼芳, 白继峰, 王文清, 雷良才, 李海英, 韩向艳, 胡跃鑫. 基于超支化聚对氯甲基苯乙烯聚合离子液体共混体系的制备与表征[J]. 中国塑料, 2022, 36(3): 40-47. |
| [13] | 张庭, 金清平, 宋仕娥, 曹南南, 邓思远. 不同腐蚀环境下FRP筋耐久性与寿命预测研究进展[J]. 中国塑料, 2022, 36(3): 75-81. |
| [14] | 郝春波, 刘万胜, 赵欣麟, 王岩, 王月. 本体ABS用橡胶国产化替代分析及评价[J]. 中国塑料, 2022, 36(3): 89-95. |
| [15] | 张瑞, 姚佳斌, 李轩, 何雪莲. 双向可逆形状记忆乙烯⁃醋酸乙烯酯共聚物的制备和性能研究[J]. 中国塑料, 2022, 36(12): 6-15. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
 京公网安备11010802034965号
京ICP备13020181号-2
京公网安备11010802034965号
京ICP备13020181号-2